
A really interesting study was reported by Nature News, and has been picking up steam around the interwebs (including Wired and Scientific American). The headlines read “Genetically modified crops pass benefits to weeds” and “weeds get unintended ‘fitness’ boost from genetic modification.” The stories are reporting on a study in New Phytologist titled “A novel EPSP synthase transgene for glyphosate resistance stimulates growth and fecundity in weedy rice (Oryza sativa) without herbicide.” Sorry, not open access. I’ve read the paper a couple times now, and the results are pretty interesting, but I’m not ready to concede the transgene is causing the effects the authors observed just yet. There are just too many questions unanswered. The authors conducted many experiments (almost none of which were repeated) and used different genetic material for different experiments. I’ve tried to organize my questions and comments below as linearly as possible, but because of the structure of the article, some bouncing around is necessary. My main concerns (with much additional detail and context below):
- The breeding methods used did not ensure enough genetic uniformity between GE and non-GE hybrids to attribute differences to the transgene as they claim.
- The authors provided very little information about the the original transformation event, and how the transformed line differed from its parent line (even after presenting data suggesting the lines had different phenotypes).
- The authors do not adequately discuss other possibilities that may explain their results, particularly with respect to the promoter they used and the impact of closely linked genes.
- Closely related traits (tiller production, panicle production, seed production) are presented as independent evidence of an increase in fitness.
- None of the experiments presented in this study were repeated.
Repetition of experiments
Repeating experiments is a requirement of nearly all reputable crop and weed science journals, particularly where the environment may significantly influence results. As an example, the Weed Research instructions to authors states:
“There should be sufficient material presented so that the information is of wider interest than just for local conditions. Thus, single experiments are unlikely to be acceptable. Research should cover sufficient temporal and spatial variation to be able to make sound generalisations. For example, evaluation of herbicide efficacy should be over more than one year at more than one site or soil type.” Weed Research Author Guidelines
The journals Weed Science, Crop Science, and most others in the field have similar requirements. The reason for this requirement is that there is inherent variability in most weed and crop research. Weeds tend to be very genetically diverse (including weedy rice; more on that later), and therefore a variable response to the growing environment is expected. It is therefore necessary to repeat experiments in time and/or space to ensure the effects are repeatable and generalizable. Repetition of experiments also decreases the chance of a Type I error (concluding there are differences when none actually exist). The only experiment in the Wang et al. paper that was repeated was the field study; but different genetic material was used in the 2009 and 2011 field studies.
Genetic material used in the study
The authors created their putative resistant rice strain by adding a maize promoter to a native rice EPSPS gene.The authors cloned the EPSPS gene from the rice cultivar ‘Minghui-63’, added a maize ubiquitin promoter, and inserted the gene into the cultivar ‘Minghui-86’. The transformed rice strain was then named ‘EP3’.
“This line was generated via Agrobacterium-mediated transformation and selfed through the T5 generation.”
Presumably, this means that the two rice strains ‘Minghui-86’ and ‘EP3’ were near-isogenic; that is, the varieties were nearly identical genetically except for the presence of the transgene. The authors imply this, but never present any data confirming the similarity of 2 rice varieties. Rather, tucked away in the supplemental information file, Table S1 actually indicates there is a rather large difference between the ‘EP3’ and ‘Minghui-86’ rice lines.
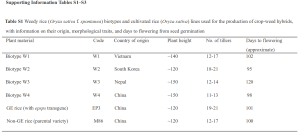
Notice the number of tillers produced by the GE rice (‘EP3’) compared to the Non-GE rice (‘Minghui-86’). The difference in tillers per plant ranges from 2 to 9; that is a rather important difference between the two lines that they are using to generate their F1 progeny. There are 2 possibilities for this difference; the most likely possibility is that the two rice varieties are not near-isogenic, and that they are genetically different enough to produce a significantly different number of tillers. The other possibility is that the two lines are near-isolines, and that the transgene in ‘EP3’ is actually responsible for the increased number of tillers. I can think of no obvious mechanism that would cause over-expression of EPSPS to result in additional tiller production. And also importantly, if the over-expression of EPSPS actually resulted in increased tiller production in the transformed ‘EP3’ line, why did the authors not explore this more fully? This seems like far more important information than the effect on weedy rice hybrids.
UPDATE (September 24, 2013): Dr. Allison Snow (a co-author on the paper) informed me a while back that they have submitted “a fairly significant correction and clarification” to their paper. The primary change can be found in Supplemental Table S1. The change is related to my point above regarding the comparison of the GE and non-GE parental lines. Here is the updated table if you would like to compare them for yourself. Basically, they indicate that a statistical test was done to confirm tiller number was indeed significantly different between the parent line and the transformed line, and provided the tiller numbers as mean+/- standard errors instead of a range of values.
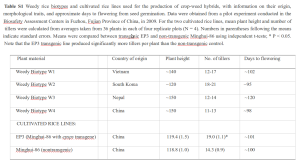
Development of cultivated x weedy rice hybrids
The authors used 4 different weedy rice biotypes collected from 4 different countries. To create the F1 hybrids (see figure below), they crossed each weedy rice biotype with either the ‘EP3’ to generate GE hybrids, or ‘Minghui-86’ to create non-GE hybrids. Because there are some notable differences (particularly in tiller production) between ‘EP3’ and ‘Minghui-86’, we shouldn’t be surprised that there are differences between the resulting F1 hybrids (also in tiller production). Therefore, I think we cannot have any confidence that the results from the 2009 field experiments, which used the F1 hybrids, provide any evidence the transgene is causing observed differences.

For all other experiments, the authors used F2 and F3 progeny. To create the F2, they allowed the GE F1 individuals to self-polinate. Since the F1 plants were hybrids between the ‘EP3’ and wild rice, all F1 progeny contained one copy of the EPSPS transgene. When the F1 plants were self-pollinated, approximately 75% of the F2 generation would be expected to contain the transgene (25% homozygous, or two copies of the gene; and 50% heterozygous, with one copy of the gene). The remaining 25% would be expected to have no copies of the transgene (thus designated non-GE F2). Likewise, they allowed the F2 plants to self pollinate, and the F3 plants again consisted of some plants with the transgene and some without. The authors then used either the F2 or F3 individuals for the remaining experiments. In the F3 generation, the authors selected only plants that were homozygous for the EPSPS transgene (either ++ or –) for the GE and non-GE F3 progeny. The authors defend their choice to use this methodology the following way:
“Crop–weed hybrids represent an excellent system for testing for fitness consequences of transgenes because the F2 and subsequent progeny segregate for the presence or absence of the transgene of interest. Thus, GE and non-GE progeny have similar genetic backgrounds, differing only in the presence of the transgene and any traits linked to the transgene insertion site.”
This seems like a pretty major overstatement to me. Certainly, since the F2 and F3 generations were a result of self-pollination, there will indeed be segregation of the novel EPSPS trait. But to say that they differ “only in the presence of the transgene and any traits linked to the transgene insertion site” seems inaccurate. The F1 generation was the result of a cross between GE rice and weedy rice, and thus ~50% of the genetics came from weedy rice. And there is a very large amount of genetic diversity in weedy rice populations. One common method for quantifying genetic diversity is the Shannon Diversity Index, which ranges from 0 to 1; zero represents a complete lack of diversity (e.g., a single genotype), and higher numbers represent a corresponding increase in genetic diversity. One recent study from China has suggested that genetic diversity in weedy rice has increased over time, and that up to 45% of total genetic diversity occurs within populations. The two most recent populations in this Chinese study had Shannon Diversity Indices of 0.397 and 0.728. By comparison, kochia (Kochia scoparia), a troublesome outcrossing weed in the US, is often considered to have very high genetic diversity. Using similar methods, kochia was reported to have Shannon Diversity Index values of 0.42 to 0.52. Now, the number of biotypes used and the number of genetic sequences analyzed can impact these numbers quite a bit, but I think we can safely conclude that there exists substantial genetic diversity within weedy rice.
So the four weedy rice populations used by Wang et al. were probably heterozygous at many loci (or at the very least the populations were a mixture of many homozygous individuals), meaning the individual plants in the GE F1 generation would have had quite a different mix of genotypes. By self-pollinating the F1 generation, the F2 population would be segregating for the transgene as the authors pointed out, but they would probably also be segregating for many other traits at the same time. The F3 generation would be segregating for a fewer number of traits, but still segregating. So it is simply incorrect to characterize the F2 and F3 generations the way the authors did:
“In the F2 and F3 generations, the only difference between GE and non-GE crop–weed progeny was the presence or absence of the inserted epsps construct (Fig. 1), as well as the selectable marker gene and any crop-specific genes that may be linked to the transgene insertion site.”
Closely linked genes
Speaking of “crop-specific genes that may be linked to the transgene insertion site,” what about that? Since the authors self-pollinated the F1 to get F2, and selfed the F2 to get the F3, each generation would be segregating for any heterozygous loci. As I mentioned above, since weedy rice is genetically diverse this could be quite a few genes still segregating at the F2 and F3 generation. But even more interesting is the bias introduced by the selection process. In the F2 and F3 generations, Wang et al. determined whether the trasngene was present or not in all progeny. They called any individuals with the transgene “GE F2”, and any individuals lacking the transgene “non-GE F2”. This selection process will cause any genes in near proximity to the transgene insertion site to be far more likely to show up in the GE progeny compared to the non-GE progeny. Likewise for the F3 progeny. If there aren’t any exceptionally interesting genes near the transgene insertion site, then this isn’t going to be a very big deal. But if there is an important gene (say, a gene that helps regulate tiller production), then this selection bias can have a very important impact on the results. Nearly all the GE F2 and F3 progeny would get this closely linked “tillering gene” from the cultivated rice parent (which has been domesticated to have good tillering characteristics). But the non-GE F1 progeny would only have about a 50% chance of receiving the “tillering gene” from the cultivated parent, and an equal likelihood of getting that gene from the weedy parent. The probability of F2 and F3 progeny getting the gene would then be dependent on whether the F1 parent had the gene. The same would go for any other genes that are located near the transgene insertion site. The authors of the paper acknowledge that this is a possibility, but discount the possibility due to what I consider a rather odd reason [emphasis mine]:
“Given the consistency of our results, we assume that overproduction of EPSPS and the downstream differences that we observed between GE plants and their non-GE counter-parts were attributable to the over-expression of the modified transgene, epsps, rather than other tightly linked genes from the cultivated parent, although this possibility cannot be ruled out entirely.”
I really don’t see how the consistency of results gives any indication of whether the results were due to the transgene or not, given their methods. There is really no way to tell from the information presented if the effect is due to the transgene or closely linked genes, regardless of how consistent their results are. If they repeated the study by inserting their gene into a different location, and then observed consistent results, then I would be more inclined to accept this reasoning. But again, none of the experiments in this study were repeated.
Another possibility related to closely linked genes is the idea that the promoter they used is actually causing closely linked genes to be over-expressed. The authors present no evidence for or against this theory, but it seems possible. The promoter they used is the maize ubiquitin promoter. Maize ubiquitin is “expressed everywhere at every developmental stage” in corn, although not equally in all tissues/growth stages. So since this promoter resulted in strong expression of EPSPS in rice, it is quite possible that it has also increased production of other products coded for by closely linked genes. Which brings up an interesting question: could the promoter from a transgene increase expression of native genes that have a positive or negative effect on fitness? Even if the results presented by Wang et al. are not due to over-expression of EPSPS as the authors claim, it is possible that the effects are due to a strong promoter being inserted in just the right place and increasing expression of some native genes nearby. But, again, the authors did not explore this possibility and did not provide any evidence for or against.
Mechanism and level of glyphosate-resistance
The authors point out correctly in the introduction that all transgenic glyphosate-resistant crops currently on the market contain a modified EPSPS gene that produces an enzyme that has a low affinity for glyphosate. Glyphosate kills plants by binding to the EPSPS and preventing it from doing its job properly. If glyphosate can’t bond to the enzyme, the plant will be resistant. The authors note that in contrast to the commercially available mechanism of resistance:
“our current study involved transgenic over-expression of an endogenous epsps gene as a mechanism for glyphosate resistance.” (Wang et al. 2013)
Over-expression of EPSPS has been demonstrated in at least 7 different crops using conventional breeding techniques, but has been unsuccessful at producing stable, commercial levels of glyphosate resistance at the whole-plant level. So it is interesting that the authors indicate they have achieved glyphosate-resistance using this method. But, notably, the authors never actually state how resistant their modified rice line is.
“These plants are resistant to glyphosate (F. Wang, unpublished data) and may be developed for commercial rice production in the future.”
They also mention that their approach of over-expressing EPSPS “can be very effective in tobacco and rice”. The reference in support of tobacco is also a little cagey about the level of glyphosate-resistance, only stating their GM tobacco lines “were tested by spraying RoundupTM with a commercial recommended dose.” The reference in support of rice was, again, “unpublished data.” Perhaps it is simply an oversight that the authors (and reviewers) of the papers were not concerned with, but it seems odd to me to say “commercial dose” instead of simply saying 840 g/ha or 250 g/ha, or whatever rate they used.
UPDATE (September 24, 2013): In a press release from Ohio State University dated 9/23/13, Dr. Snow states “This is a relatively new way to get a trait into a crop: taking the plant’s own gene and ramping it up,” But this approach is not new, and was was first published over 25 years ago in both petunia and arabidopsis. </UPDATE>

Anyone familiar with the Roundup label will understand that “commercial recommended dose” provides very little information. Depending on the weed and crop you’re spraying, the “recommended dose” can range nearly 10-fold, from 11 fluid ounces per acre (0.38 lbs ae/A) to 3.3 quarts per acre (3.7 lbs ae/A). In order to commercialize this trait, the rice would probably need to withstand a minimum of 1.5 lbs ae/A glyphosate under field conditions. But we are given no evidence other than references to “F. Wang, unpublished data” supporting that the parental rice line is resistant to glyphosate. The tobacco paper shows some seemingly convincing photos (See Figure 3 here). I’ve got similar photos of weeds sprayed with glyphosate ; but all of the lambsquarters in my photos to the right were susceptible to commonly used rates of glyphosate (0.75 lbs ae/A) in the field. So without knowing the rate applied, photos don’t tell us much. Without this data, it is difficult to assess whether the trait would ever even be released into the wild, making its effect on weedy rice a moot point.
What if it is true?
For the reasons I described above, I don’t think the authors present convincing evidence that the transgene has resulted in fitness gains in the weedy rice hybrids. But it is certainly plausible, and would be interesting to have some more rigorously controlled studies to investigate the possibility. But it seems to me that the authors of this paper have put the cart before the horse; they are studying the effect of a transgene on weedy hybrids, without first investigating the impact their transformation had on the parental line. Although they claim to have tested and confirmed glyphosate-resistance (although only “unpublished data” supporting this), they don’t report any studies on whether the transformed ‘EP3’ line is different than its parent line in other ways. The tiller production data hidden away in the supplemental information Table S1 indicates there is a difference between parent and transformed line. Could it be possible that this difference was caused by the transformation? One of the results of the 2011 field studies was also that tiller production was greater in the GE compared to non-GE F2 progeny. Increased tiller production is probably responsible for the increased panicle number and seed production the authors also observed. So is it possible that either a strong promoter or over-expression of EPSPS could have actually increased tiller production in the transformed line, and passed that difference to the weedy hybrids? This wouldn’t be the first time this has been done. Monsanto’s “Roundup Ready 2 Yield” trait in soybean is described the following way:
Monsanto developed Genuity Roundup Ready 2 Yield soybeans through extensive gene mapping that identified specific DNA regions in soybeans that have a positive impact on yield. Using advanced insertion and selection technologies, the Genuity® Roundup Ready 2 Yield® gene is situated in one of these high-yield DNA regions to help deliver high-yield potential.
If the transformed rice line had its yield potential increased by the transgenic event (regardless of the mechanism), this would be an important discovery. One of the common criticisms of GMO crops is that they don’t increase yield of the crops per se, but only have an impact in the presence of the pest. If this GE trait were commercialized, and the tiller and yield differences are really due to the transformation event, this criticism would no longer hold much weight. Instead of being touted as a problem when it gets into weedy rice, the GE trait could actually provide a benefit to farmers even if it is never sprayed with glyphsoate. But I think more research is required before we can conclude much of anything.
[Updated August 22, 9:05 AM to add information on Roundup Ready 2 Yield soybean suggested by Craig Reid]


Comments are closed.